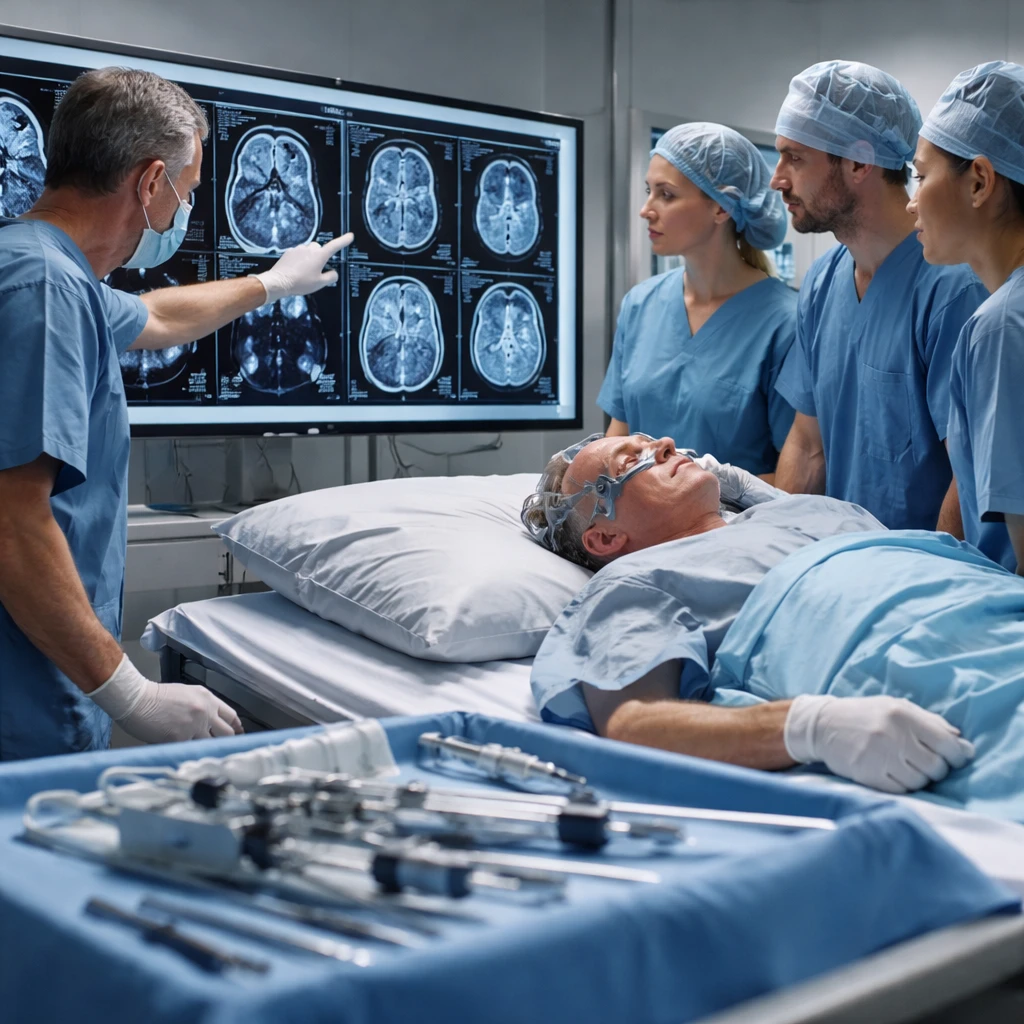
Summary: Alpha Tau Medical Ltd. (NASDAQ:DRTS) reported interim REGAIN trial results that prompted an 18% rise in its shares in premarket trading Monday. The Jerusalem-based company said two of the first three recurrent glioblastoma patients treated with Alpha DaRT had complete response on follow-up MRIs, while the third patient achieved stable disease with measurable tumor shrinkage.
Trial outcomes and safety review
The company disclosed that two patients experienced complete response, defined as total disappearance of all enhancing tumor lesions on multiple magnetic resonance imaging scans since treatment. The third patient had stable disease with a 30% decrease in tumor dimension. These results are from the initial trio of patients enrolled in the U.S. REGAIN study.
The interim safety analysis was conducted at the request of the U.S. Food and Drug Administration and encompassed patients treated in December 2025, February 2026 and March 2026, with a data cutoff of May 3, 2026. The analysis recorded one grade 3 serious adverse event - a seizure accompanied by temporary paralysis - which resolved after administration of steroids. The company reported no unanticipated serious adverse events. As of the cutoff date, none of the three patients had shown local or distant recurrence, nor residual symptoms attributable to the procedure.
Clinical setting and delivery
All three patients were treated at The Ohio State University Comprehensive Cancer Center by a multidisciplinary team led by Principal Investigator Joshua D. Palmer. The procedure used a delivery approach developed specifically for intracranial application of Alpha DaRT.
Context on glioblastoma
Alpha Tau noted the difficult prognosis associated with glioblastoma, citing figures from the National Brain Tumor Society that place the estimated average survival at roughly eight months, with approximately 14,000 new U.S. diagnoses annually and a five-year survival rate under 10%.
Company response and next steps
CEO Uzi Sofer said the interim readout supports the company’s strategy to develop new treatment options for glioblastoma, a cancer type where recurrence is common and median survival after recurrence is usually measured in months. Alpha Tau indicated it plans to continue enrolling and treating patients in the REGAIN trial while awaiting FDA confirmation following the agency’s review of the submitted safety report.
This interim dataset is limited to the three treated patients referenced above and to the safety follow-up through the May 3, 2026 cutoff; the company will proceed with additional patient treatment and regulatory engagement as described.